Project Management Services
Home  Solutions
Solutions  Development
Development


Project Management Services
Home  Solutions
Solutions  Development
Development
Driving Excellence in Pharma Projects
At PI Health Sciences, we specialize in pharmaceutical project management that ensures precision, speed, and seamless execution throughout the drug discovery and development lifecycle. With an experienced team and advanced tools, we deliver customized, high-quality outcomes tailored to client needs across the global pharmaceutical landscape.

Our Project Management Model
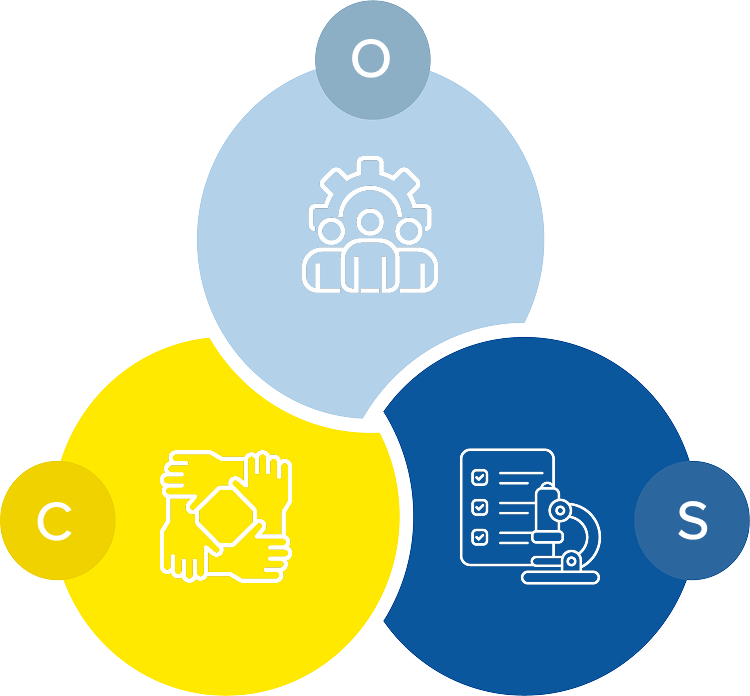
Operation Strategy:
- Expert Leadership: PMP & Agile-certified in pharmaceutical project management
- Agile Workflows: Seamless cross-functional collaboration for flexibility
- Smart Tracking: Tools like Kytes ensure real-time project visibility
Scientific Excellence:
- Data-Driven Insights: Leveraging advanced analytics and AI-driven tools.
- Regulatory Adherence: Strict compliance with global quality and regulatory standards.
- Domain Expertise: Deep scientific knowledge across cross-functional teams.
Communication & Collaboration:
- Customer-Centric Approach: Alliance-driven partnerships for seamless collaboration.
- Cloud-Based Infrastructure: Enabling real-time data sharing and project alignment.
- Transparent Updates: Regular reporting and proactive communication.
Why Partner With Us?
Integrated Solutions
Comprehensive management from discovery to development.

Experienced Team
Certified project managers with proven expertise.

Cutting-Edge Tools
Advanced platforms for tracking and analytics.

Global Standards
Adherence to quality and regulatory requirements.

Client Focused
Flexible and collaborative approach ensuring project success.
Facilities & Capabilities

Project Management Tools
Kytes and cloud-based collaboration platforms.

Analytics Integration
AI and data-driven insights for strategic planning.

Global Alignment
Strategies tailored to meet international standards.
Contact Us
Reach out today to discuss how our tailored, integrated drug discovery expertise can accelerate your program’s success.
