Preclinical Services
Integrated Preclinical Services to Enable Confident Clinical Advancement
At PI Health Sciences, our integrated Preclinical Services are designed to seamlessly bridge late-stage discovery with regulator-aligned clinical development. We generate the pharmacokinetic, pharmacodynamic, biomarker, and safety datasets required to support first-in-human studies and downstream regulatory submissions, while proactively reducing late-stage development risk.
Our preclinical model integrates DMPK, in vitro and in vivo ADME, translational biomarker science, and expert-led safety assessment within a single scientific and operational framework. This approach ensures that exposure, biological responses, and safety margins are evaluated holistically rather than in isolation, enabling informed go or no-go decisions and efficient advancement toward the clinic.
By combining predictive modelling, mechanistic insight, and GLP-aligned study strategies, we support programs from late discovery through IND and CTA readiness. Our multidisciplinary teams collaborate closely with sponsors, CRO partners, and evolving regulatory expectations to deliver scientifically robust, decision-enabling, and submission-ready data.
Our Services
Preclinical Studies
Preclinical toxicology services for drug discovery should emphasize early decision-making, IND-enabling safety, and seamless transition from discovery to development.
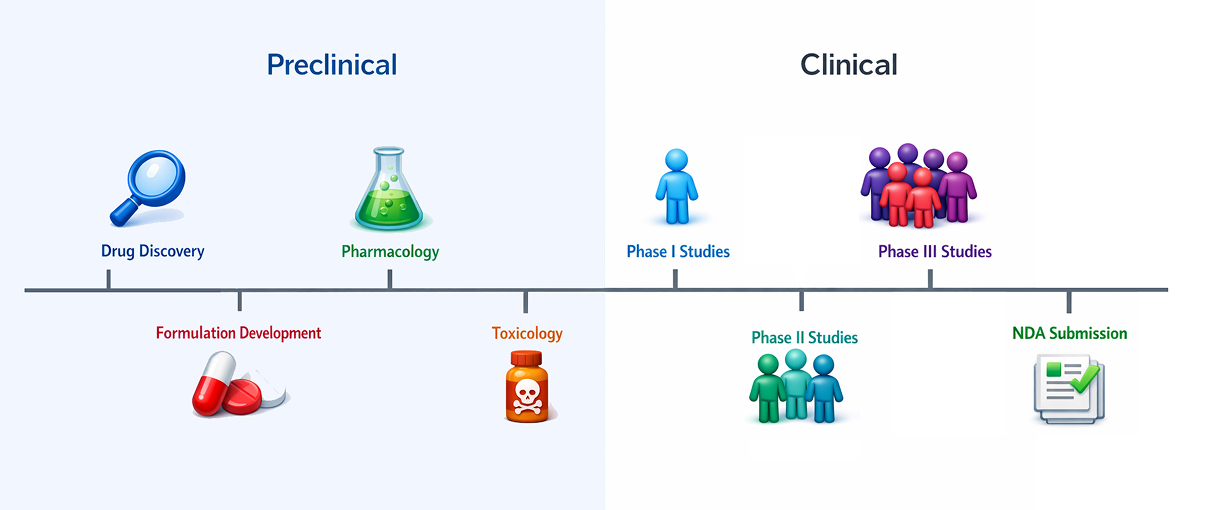
Early Discovery-Aligned Toxicology
- Non-GLP exploratory tox studies (dose-range finding, MTD, target organ identification) to quickly flag high-risk candidates.
- Mechanistic and investigative toxicology (biomarkers, histopathology, in vitro liability assays) to understand and mitigate toxicity drivers.
- Integrated PK/TK and exposure–response analyses to guide safe and scientifically justified dose selection for subsequent studies.
Patient selection hypothesis, biomarkers and human dose projection
A key translational objective in preclinical development is to predict the human active dose range corresponding to exposure levels required for
pharmacological activity and efficacy in preclinical models.
PIHS applies stage-gate criteria developed by key opinion leaders
(KOLs) to strategically guide project teams and maximize the probability of
achieving clinical proof-of-concept. In collaboration with external partners, we develop strategies for human pharmacokinetic (PK) prediction, including estimation of clearance (CL) and bioavailability, to enable accurate projection of a safe and efficacious first-in-human (FIH) dose.
Our approaches for biomarker validation and testing include quantitative gene expression, multicolor
flow cytometry, ELISA, Flourospot, western blotting and next generation sequencing. Our collaboration
with local medical centres gives us access to patient derived tissues and biopsy samples which helps
validation of known disease related biomarkers.
Biomarker analysis
- Test systems
- Peripheral whole blood,
- Disease-relevant organs and tissues (ex vivo),
- Patient derived liquid biopsies
- Patient derived cancer biopsies,
- Bronchoalveolar lavage fluid (BALF)
- Synovial fluid
- Methodologies
- Soluble biomarker analysis by ELISA
- Cell-based biomarker by FACS and Western Blot
- Gene expression profiling by RT-PCR
- Tumor-infiltrating lymphocyte (TIL) analysis in human and murine tumor tissues using flow cytometry.
Talk to Our Experts
Translational Biology
De-Risking Human Translation Through Biomarker-Driven Decision Science
Translational Biology at PI Health Sciences serves as the critical scientific bridge between in vitro
discovery and clinical proof-of-concept. Our focus is to ensure that the right biological hypotheses,
patient populations, and dose strategies are established before clinical entry, reducing uncertainty and
late-stage failure. By integrating human-relevant models, biomarker science, and quantitative
pharmacology, we enable confident translation of preclinical findings into clinically actionable
insights
Our translational biology platform is designed to define exposure–response relationships, validate
pharmacodynamic and safety biomarkers, and support human dose projection. We leverage primary human
tissues, patient-derived systems, and high-dimensional molecular readouts to ensure that biological
signals observed preclinically remain relevant in the clinical setting. This approach strengthens
First-in-Human dose justification, patient stratification strategies, and regulatory narratives.
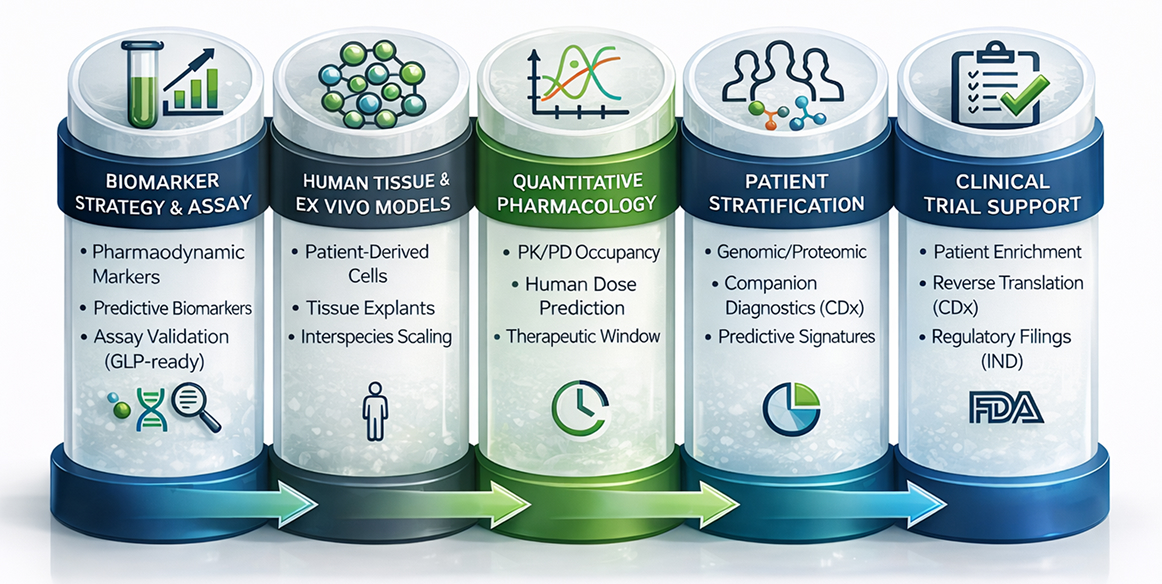
How Our Approach Is Different
Translational Biology at PI Health Sciences is embedded directly within discovery, DMPK, and safety workflows rather than functioning as a downstream validation layer. This enables reverse translation, where unexpected biological outcomes immediately inform molecular design, biomarker refinement, or dose strategy, accelerating learning cycles and preserving program momentum
Core Design Principles
- Biomarker strategies designed to measure target engagement, pharmacology, and emerging safety signals
- Use of human primary cells, ex vivo tissues, organoids, and patient-derived explants for improved translatability
- Integrated PK, PD, and target occupancy modelling to support human dose projection
- Quantitative pharmacology approaches linking exposure to biological response
- Patient stratification and precision medicine frameworks to guide clinical positioning
- Continuous alignment with discovery, DMPK, and regulatory expectations
Talk to Our Experts
Toxicology Studies
Decision-Ready Safety Science to Enable Confident Clinical Entry
Toxicology at PI Health Sciences is purpose-built to support modern drug discovery programs with
phase-appropriate, decision-driven safety assessment. Our toxicology capabilities are designed to
identify liabilities early, guide candidate selection, and generate regulator-aligned safety data that
supports progression to First-in-Human studies with confidence
We deliver both exploratory and IND-enabling toxicology through integrated study design,
combining safety endpoints with toxicokinetics, bioanalysis, and mechanistic insight. By aligning
toxicology strategy with discovery timelines and translational objectives, we ensure that safety data
actively informs development decisions rather than acting as a late-stage hurdle.
How Our Approach Is Different
Toxicology at PI Health Sciences is discovery-aligned by design. Rather than treating safety assessment as a standalone compliance function, our toxicologists work closely with DMPK scientists, biologists, and external partners to ensure that exposure, mechanism, and toxicity are interpreted together. This integrated model enables faster risk identification, smarter study sequencing, and stronger IND-ready safety packages
Core Design Principles
- Discovery-aligned exploratory toxicology to flag liabilities early
- Non-GLP dose range finding and mechanistic studies to guide candidate selection
- GLP-compliant repeat-dose toxicology aligned with global regulatory guidance
- Integrated PK, TK, and exposure–response interpretation
- Bioanalysis, DMPK, and formulation support embedded within tox programs
- Strategic study design and documentation to support IND and CTA submissions
Talk to Our Experts
Capabilities


Integrated DMPK and Translational Preclinical Strategy
An integrated DMPK, biomarker, and safety framework aligns preclinical data early to enable clear exposure, dosing, and regulatory decisions without downstream rework.
In Vivo Pharmacokinetics and Pharmacodynamics
Multi-species PK, tissue distribution, dose proportionality, and PK-PD correlation studies to support translational interpretation and inform clinical dose selection strategies.
Translational Biomarker and Exposure-Response Science
ELISA, flow cytometry, RT-PCR, multiplex cytokine panels, and advanced molecular assays to directly link systemic exposure with biological response, pharmacology, and emerging safety signals.
Human PK Prediction and Dose Modelling
PBPK and compartmental modelling approaches to support first-in-human dose selection, exposure projections, and clinical study design, reducing uncertainty at clinical entry.
Regulatory-Aligned Safety and Study Execution
Non-GLP dose range finding and GLP-compliant safety studies planned and executed by experienced Study Directors, with protocols aligned to FDA, EMA, OECD, and global regulatory expectations.

Submission-Ready Data and Regulatory Confidence
Studies are designed, executed, and interpreted with IND, CTA, and NDA submissions in mind, ensuring coherent datasets, defensible dose rationales, and consistent regulatory positioning across programs.
Frequently asked questions
We’re here to help with any questions you have about our plans, supported features, and how our model works.
How does PI Health Sciences integrate preclinical activities within a single program framework?
PI Health Sciences plans DMPK, translational biology, PK modelling, and safety studies as a unified preclinical program. Data generated at each stage directly informs subsequent study design, including species selection, dose levels, exposure targets, and safety margins, ensuring continuity and regulatory coherence.
How is First-in-Human dose selection supported during preclinical development?
First-in-Human dose selection is supported through integrated human PK prediction, exposure–response modelling, and safety margin assessment. In silico modelling and PK-guided approaches are combined with nonclinical data to generate regulator-aligned, scientifically justified dose rationales.
What types of preclinical safety studies are supported?
PI Health Sciences supports non-GLP dose range finding and GLP-compliant safety studies designed in alignment with global regulatory guidelines. Studies are led by experienced scientific teams and delivered with submission-ready reporting to avoid downstream delays.
How does PI Health Sciences ensure translational relevance across preclinical studies?
Translational relevance is ensured through disease-relevant biomarkers, integrated PK interpretation, and exposure-linked biological readouts that connect preclinical findings to downstream clinical hypotheses.
Contact Us
Connect with PI Health Sciences to explore how our pre-clinical services de-risk development and prepare therapeutic candidates for IND submission with precision and scientific rigor.
